Prof Martin Knight
BEng, MSc, PhD, FHEA
Research Overview
Organ-on-a-Chip, Mechanobiology, Primary Cilia, Cartilage, Bone, Cancer, Inflammation, Cardiovascular diseases
My research concerns the area of mechanobiology or how living cells and tissues ‘sense and respond’ to mechanical forces. These fundamental biological processes are involved in both health and disease for many tissues. As a result mechanobiology has potential application in various medical therapies as well as in the development of organ-on-a-chip models for pre-clinical testing. The group are examining the role of mechanobiology and developing organ-chip models for a variety of conditions including ageing, cancer, inflammation, kidney disease, atherosclerosis and arthritis.
Much of my mechanobiology research aligns with the following areas:
Organ-on-a-Chip Technology

I was co-director of the UK organ-on-a-chip technologies Network led by Prof Hazel Screen and funded by MRC, EPSRC and BBSRC. I am Director of the Queen Mary & Emulate Organs-on-chips Centre the first of its kind in the UK providing access to state-of-the-art organ-chip technology and validated organ-chip models. With Prof Hazel Screen we have set up the cross-faculty Centre for Predictive in vitro Models and associated PhD Training.
My research activity is focussed on the development of organ-on-a-chip in vitro models with a particular focus on incorporation of appropriate biomechanical stimuli to replicate the physiological and pathological environment. Research projects funded by MRC, EPSRC, BBSRC, EU, NC3Rs and CRUK are examining the development of organ-chips for study of musculoskeletal disease, polycystic kidney disease, atherosclerosis and bone metastasis (see fundng for details)
I am actively seeking collaboration with academic and industry partners as well as incoming PhD students and research fellows through schemes such as the Marie Skłodowska-Curie Fellowships.
Primary Cilia Structure and Function
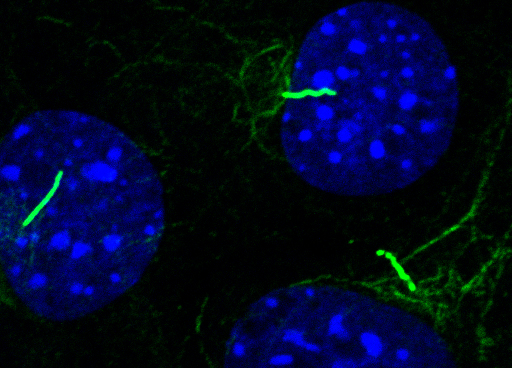 A particular focus of my research involves the role of primary cilia in mechanobiology. These fascinating cellular structures have been largely ignored, but have recently been shown to be involved in mechanosignalling although the mechanisms are not yet clear. Furthermore mechanical forces regulate the expression of cilia which in turn modulates other signalling pathways. My group are examining the relationship between cilia structure and function and how this is influenced by physicochemical stimuli. Our studies have shown how regulation of cilia length controls hedgehog signalling (Thompson et al. 2014; 2015; Prodromou et al. 2012), wnt signalling (McMurray et al. 2014), and growth factor signalling (Dalbay et al. 2015). Our group published the first paper showing that primary cilia are required for cartilage mechanotransduction (Wann et al. 2012a) and we have also shown that primary cilia regulate inflammatory signalling in response to cytokines such as interleukin-1 (Wann et al. 2012b; 2013; 2014).
A particular focus of my research involves the role of primary cilia in mechanobiology. These fascinating cellular structures have been largely ignored, but have recently been shown to be involved in mechanosignalling although the mechanisms are not yet clear. Furthermore mechanical forces regulate the expression of cilia which in turn modulates other signalling pathways. My group are examining the relationship between cilia structure and function and how this is influenced by physicochemical stimuli. Our studies have shown how regulation of cilia length controls hedgehog signalling (Thompson et al. 2014; 2015; Prodromou et al. 2012), wnt signalling (McMurray et al. 2014), and growth factor signalling (Dalbay et al. 2015). Our group published the first paper showing that primary cilia are required for cartilage mechanotransduction (Wann et al. 2012a) and we have also shown that primary cilia regulate inflammatory signalling in response to cytokines such as interleukin-1 (Wann et al. 2012b; 2013; 2014).
Through understanding these fundamental behaviours and the effect of physicochemical stimuli we hope to have future impact in the development of 'ciliotherapies' for treatment of disease and injury. Recent work has screened over 17,000 FDA-approved drugs to identify compounds that regulate cilia expression and may therefore be suitable for drug repurposing or as a means to identify novel therapeutic targets.
I helped set up the UK Cilia Network bringing together researchers with a common interest in cilia.

