Prof Yi Sui
PhD, MInstP, Fellow HEA
Research Overview
Biofluids and Cell biomechanics, Multiphase flows, Transport phenomena, Microfluidics, High-speed imaging, real-time AI
The research in the Intelligent Biofluid Mechanics Group mainly concerns modelling, simulation and characterisation of suspended biological cells. We develop high-fidelity mechanical models, computational methods, real-time AI algorithms, and high-speed imaging and data streaming systems, and integrate them to build new tools with unparalleled performance for label-free cell phenotyping and sorting.
Recent research highlights
1. A computational study of cell membrane damage and intracellular delivery in a cross-slot microchannel (front cover article in Soft Matter)
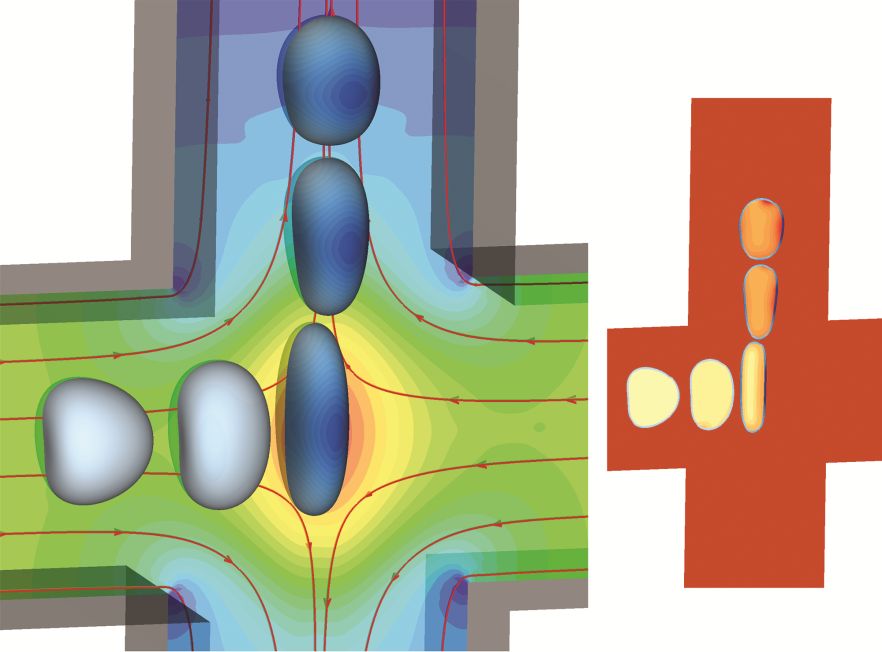
We propose a three-dimensional computational framework to simulate the flow-induced cell membrane damage and the resulting enhanced intracellular mass transport in a cross-slot microchannel. We model the cell as a liquid droplet enclosed by a viscoelastic membrane and solve the cell deformation using a well-tested immersed-boundary lattice-Boltzmann method. The cell membrane damage, which is directly related to the membrane permeability, is considered using continuum damage mechanics. The transport of the diffusive solute into the cell is solved by a lattice-Boltzmann model. After validating the computational framework against several benchmark cases, we consider a cell flowing through a cross-slot microchannel, focusing on the effects of the flow strength, channel fluid viscosity and cell membrane viscosity on the membrane damage and enhanced intracellular transport. Interestingly, we find that under a comparable pressure drop across the device, for cells with low membrane viscosity, the inertial flow regime, which can be achieved by driving a low-viscosity liquid at a high speed, often leads to much larger membrane damage, compared with the high-viscosity low-speed viscous flow regime. However, the enhancement can be significantly reduced or even reversed by an increase of the cell membrane viscosity, which limits cell deformation, particularly in the inertial flow regime. Our computational framework and simulation results may guide the design and optimisation of microfluidic devices, which use cross-slot geometry to disrupt cell membranes to enhance intracellular delivery of solutes.
2. A computational model for the transit of cancer cells through a constricted microchannel. (Biomechanics and Modelling in Mechanobiology)
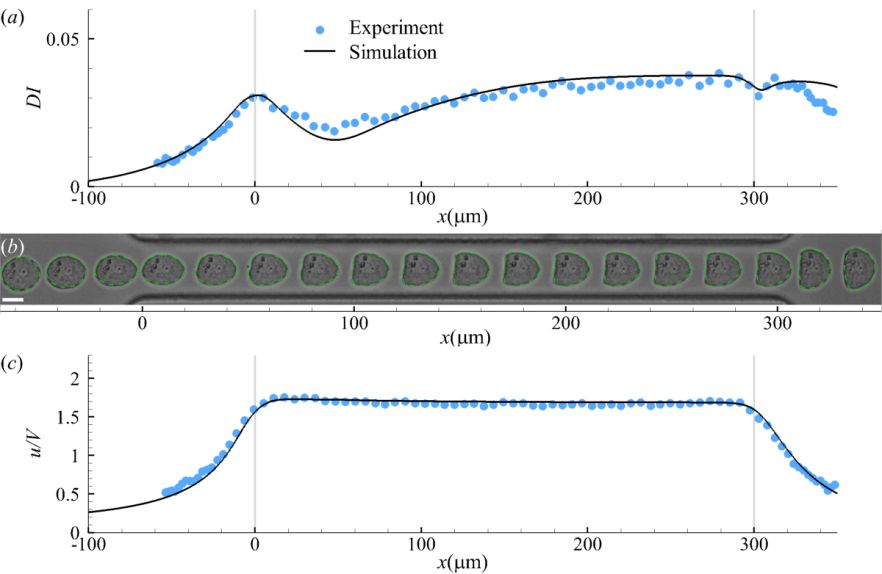
The dynamics of cancer cells flowing in microchannels is a fundamental problem that lies in the heart of numerous biomedical applications. Examples include but not limit to label-free microfluidic devices to enrich circulating tumor cells from the blood samples of cancer patients, bioprinting of tumor cells to build 3D tumor models.
In this project we build a three-dimensional computational framework to simulate the transient deformation of suspended cancer cells flowing through a constricted microchannel. We find that the classical Skalak’s law can accurately predict the steady deformation of the cancer cell in the straight channel, however, for cell transient deformation in the constriction region, excellent agreement with the experiment can only be achieved by employing a viscoelastic cell membrane model with the membrane viscosity depending on its mode of deformation (shear versus elongation)
3. A method for real‑time mechanical characterisation of microcapsules (Biomechanics and Modelling in Mechanobiology)
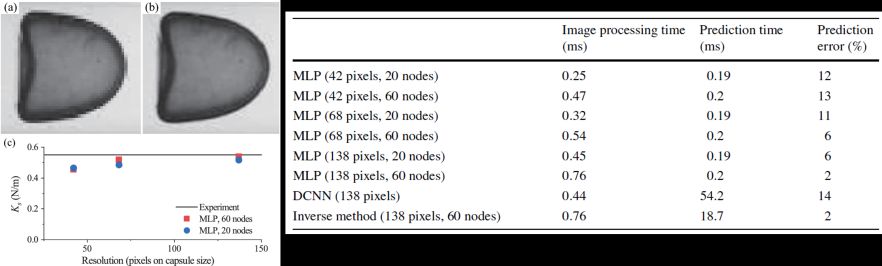
We develop a novel multilayer perceptron (MLP)-based machine learning (ML) approach, for real-time simultaneous predictions of the membrane mechanical law type, shear and area-dilatation moduli of microcapsules, from their camera-recorded steady profiles in tube flow. By MLP, we mean a neural network where many perceptrons are organised into layers. A perceptron is a basic element that conducts input–output mapping operation. We test the performance of the present approach using both simulation and experimental data. We find that with a reasonably high prediction accuracy, our method can reach an unprecedented low prediction latency of less than 1 millisecond on a personal computer. That is the overall computational time, without using parallel computing, from a single experimental image to multiple capsule mechanical parameters. It is faster than a recently proposed convolutional neural network-based approach by two orders of magnitude, for it only deals with the one-dimensional capsule boundary instead of the entire two-dimensional capsule image. Our new approach may serve as the foundation of a promising tool for real-time mechanical characterisation and online active sorting of deformable microcapsules and biological cells in microfluidic devices.
4. Transient deformation of a viscoelastic capsule flowing through a cross-slot microchannel (Journal of Fluid Mechanics)
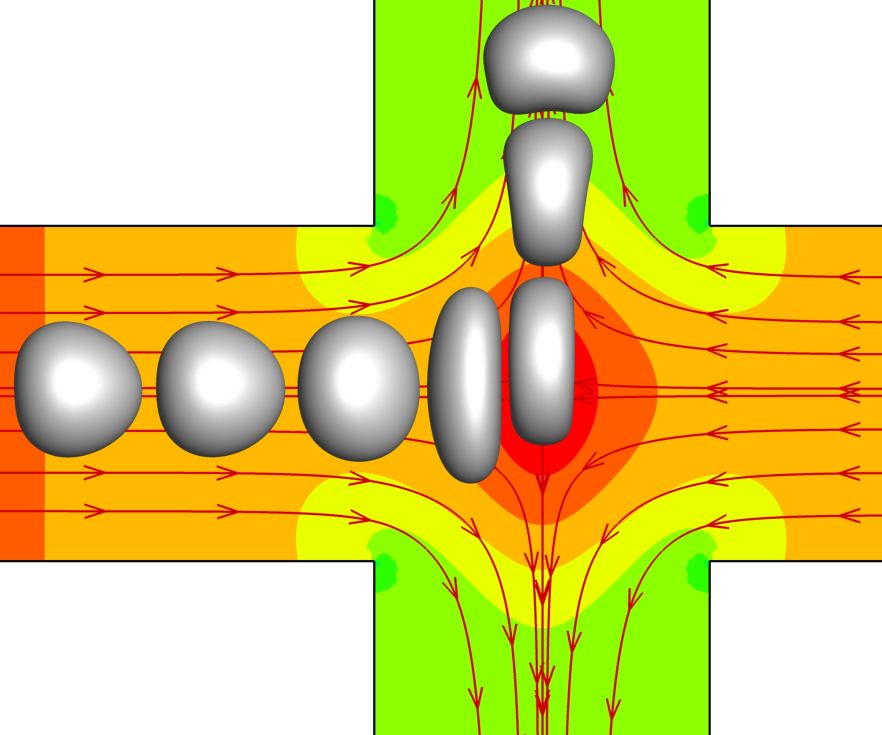
With an immersed-boundary lattice-Boltzmann method, we consider the transit of a three-dimensional initially spherical capsule with a viscoelastic membrane through a cross-slot microchannel. The capsule is released with a small initial off-centre distance in the feeding channel, to mimic experiments where capsules or cells are not perfectly aligned to the centreline. Our main objective is to establish the phase diagram of the capsule's deformation modes as a function of the flow inertia and capsule membrane viscosity. We mainly find three deformation modes in the channel cross-slot. For a capsule with low membrane viscosity, a quasi-steady mode occurs at low Reynolds numbers (Re), in which the capsule can reach and maintain a steady ellipsoidal shape near the stagnation point, for a considerable time period. With Re increasing to 20, an overshoot-retract mode is observed. The capsule deformation oscillates on an inertial-elastic time scale, suggesting that the dynamics is mainly driven by the balance of the inertial and membrane elastic forces. The membrane viscosity slows down the capsule deformation and suppresses the overshoot-retract mode. A capsule with high membrane viscosity undergoes a continuous-elongation mode, in which its deformation keeps increasing during most of its journey in the channel cross-slot. We summarise the results in phase diagrams, and propose a scaling model which can predict the deformation modes of a viscoelastic capsule in the inertial flow regime. We also discuss implications of the present findings to practical experiments for mechanical characterisation of capsules or cells.
5. A neural network-based algorithm for high-throughput characterisation of viscoelastic properties of flowing microcapsules (back cover article in Soft Matter)
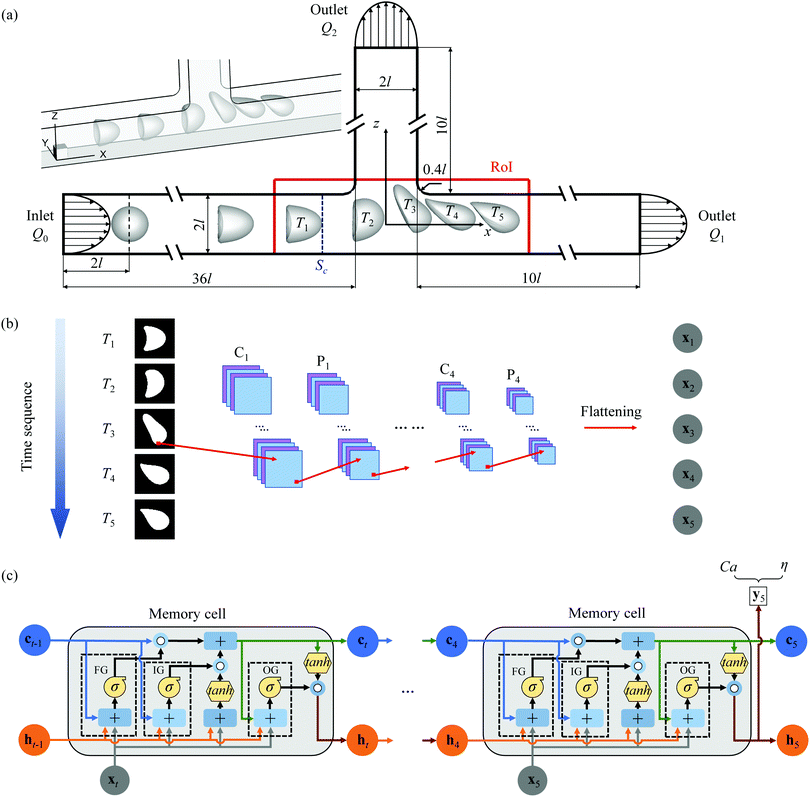
Microcapsules, consisting of a liquid droplet enclosed by a viscoelastic membrane, have a wide range of biomedical and pharmaceutical applications and also serve as a popular mechanical model for biological cells. In this study, we develop a novel high throughput approach, by combining a machine learning method with a high-fidelity mechanistic capsule model, to accurately predict the membrane elasticity and viscosity of microcapsules from their dynamic deformation when flowing in a branched microchannel. The machine learning method consists of a deep convolutional neural network (DCNN) connected by a long short-term memory (LSTM) network. We demonstrate that with a superior prediction accuracy the present hybrid DCNN-LSTM network can still be faster than a conventional inverse method by five orders of magnitude, and can process thousands of capsules per second. We also show that the hybrid network has fewer restrictions compared with a simple DCNN.
6. Path selection of a train of spherical capsules in a branched microchannel (front cover article in Journal of Fluid Mechanics)
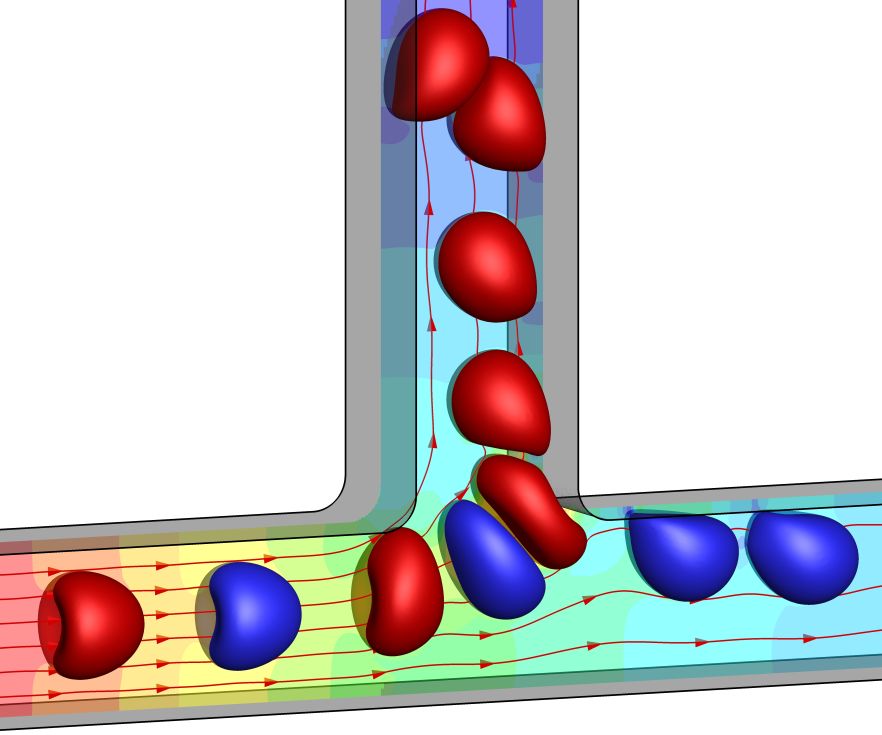
Microfluidic systems consisting of a square microchannel with an orthogonal side branch are promising tools to enrich or sort suspensions of deformable capsules. To allow their operating control, we numerically consider a train of initially spherical identical capsules, equally spaced along the axis of the feeding channel. The capsules have a strain-hardening membrane, an internal fluid viscosity identical to that of the external fluid and a size comparable to that of the channel. We study the influence of the interspacing on the capsule path selection at the channel bifurcation using a three-dimensional immersed boundary–lattice Boltzmann method. Our objectives are to establish a phase diagram and identify the critical interspacing above which hydrodynamic interaction between capsules no longer affects their path selection. We find two main regimes. At low interspacing, strong capsule interaction leads to an unsteady regime for which the capsule path selection follows either a periodic or a disordered state. Above a critical initial interspacing dct, a steady regime is achieved where interaction between capsules is too weak to affect their path selection. The capsules then follow an identical steady trajectory. We find that the dependence of the interspacing dct, normalised by the capsule radius, on the flow split ratio falls onto a universal curve regardless of the flow strength, capsule size and membrane shear elasticity. We also compare the path selection of a capsule train with that of a two-capsule system, and discuss applications of the present results in controlling capsule trains in microfluidic suspension enrichment devices.
7. A fate-alternating transitional regime in contracting liquid filaments (Journal of Fluid Mechanics)
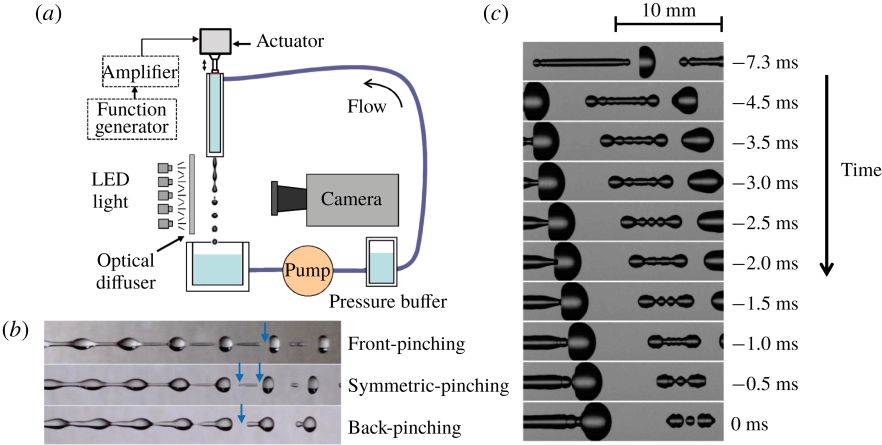
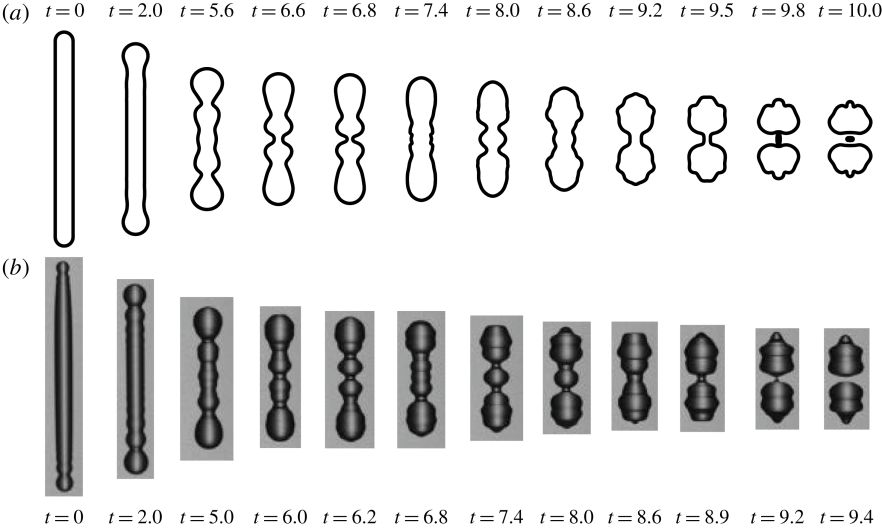
The fate of a contracting liquid filament depends on the Ohnesorge number, the initial aspect ratio and surface perturbation. Generally, it is believed that there exists a critical aspect ratio Γc such that longer filaments break up and shorter ones recoil into a single drop. Through computational and experimental studies, we report a transitional regime for filaments with a broad range of intermediate aspect ratios, where there exist multiple Γc thresholds at which a novel breakup mode alternates with no-break mode. We develop a simple model considering the superposition of capillary waves, which can predict the complicated new phase diagram. In this model, the breakup results from constructive interference between the capillary waves that originate from the ends of the filament.

