Research
The role of primary cilia in tendinopathy
| Funding source(s): | Institute of Bioengineering EPSRC PhD Studentship |
| | Start: 10-10-2013 / End: 20-06-2017 |
| Directly incurred staff: | Dan Rowsen |
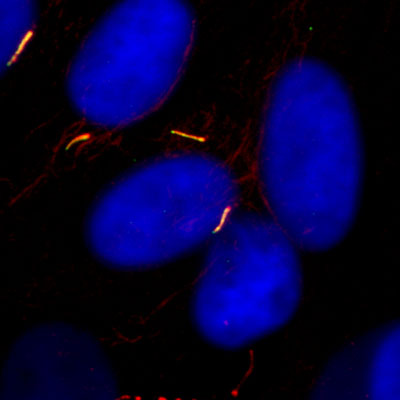 Tendon is mechanosensitive, maintaining tissue health in response to applied loads. Overload is a key contributor to the development of tendon pathologies, know as tendinopathies; a range of highly debilitating and increasingly prevalent conditions2-3. However, the mechanisms associated with tendinopathy development remain unclear. Current evidence supports a combined mechanical and cellular pathway, with early structural damage initiating a catabolic cell response. Primary cilia consist of a single microtubule axoneme, 2-4µm in length, which projects into the extracellular environment and regulates fundamental signalling pathways including mechanotransduction, hedgehog (Hh) and Wnt signalling which are important in tissue development, health and disease. Although primary cilia have been identified in tendon, their precise function is unknown. Recent studies of cilia in cartilage indicate that they are essential for tissue development and mechanotransduction mediated matrix synthesis. Furthermore cilia may play a major role in the aetiology of osteoarthritis (OA) through upregulation of Hh signalling8 and alterations in hypoxia inducible factor (HIF) expression (un-published data). Work from Knight’s group also suggests that physicochemical changes in the OA microenvironment, including mechanical loading and inflammatory cytokines, disrupt primary cilia structure and associated signalling pathways. As a tissue damaged through injurious overload, it is reasonable to hypothesise that similar cilia mediated mechanotransduction pathways may be present in tendon. This studentship will test the overall hypothesis that injurious mechanical loading influences primary cilia structure & function, creating a catabolic cell response & leading to the development of tendinopathy. This work is being conducted by Daniel Rowson funded on an EPSRC PhD studentship awarded via the Institute of Bioengineering at QMUL.
Tendon is mechanosensitive, maintaining tissue health in response to applied loads. Overload is a key contributor to the development of tendon pathologies, know as tendinopathies; a range of highly debilitating and increasingly prevalent conditions2-3. However, the mechanisms associated with tendinopathy development remain unclear. Current evidence supports a combined mechanical and cellular pathway, with early structural damage initiating a catabolic cell response. Primary cilia consist of a single microtubule axoneme, 2-4µm in length, which projects into the extracellular environment and regulates fundamental signalling pathways including mechanotransduction, hedgehog (Hh) and Wnt signalling which are important in tissue development, health and disease. Although primary cilia have been identified in tendon, their precise function is unknown. Recent studies of cilia in cartilage indicate that they are essential for tissue development and mechanotransduction mediated matrix synthesis. Furthermore cilia may play a major role in the aetiology of osteoarthritis (OA) through upregulation of Hh signalling8 and alterations in hypoxia inducible factor (HIF) expression (un-published data). Work from Knight’s group also suggests that physicochemical changes in the OA microenvironment, including mechanical loading and inflammatory cytokines, disrupt primary cilia structure and associated signalling pathways. As a tissue damaged through injurious overload, it is reasonable to hypothesise that similar cilia mediated mechanotransduction pathways may be present in tendon. This studentship will test the overall hypothesis that injurious mechanical loading influences primary cilia structure & function, creating a catabolic cell response & leading to the development of tendinopathy. This work is being conducted by Daniel Rowson funded on an EPSRC PhD studentship awarded via the Institute of Bioengineering at QMUL.
 Tendon is mechanosensitive, maintaining tissue health in response to applied loads. Overload is a key contributor to the development of tendon pathologies, know as tendinopathies; a range of highly debilitating and increasingly prevalent conditions2-3. However, the mechanisms associated with tendinopathy development remain unclear. Current evidence supports a combined mechanical and cellular pathway, with early structural damage initiating a catabolic cell response. Primary cilia consist of a single microtubule axoneme, 2-4µm in length, which projects into the extracellular environment and regulates fundamental signalling pathways including mechanotransduction, hedgehog (Hh) and Wnt signalling which are important in tissue development, health and disease. Although primary cilia have been identified in tendon, their precise function is unknown. Recent studies of cilia in cartilage indicate that they are essential for tissue development and mechanotransduction mediated matrix synthesis. Furthermore cilia may play a major role in the aetiology of osteoarthritis (OA) through upregulation of Hh signalling8 and alterations in hypoxia inducible factor (HIF) expression (un-published data). Work from Knight’s group also suggests that physicochemical changes in the OA microenvironment, including mechanical loading and inflammatory cytokines, disrupt primary cilia structure and associated signalling pathways. As a tissue damaged through injurious overload, it is reasonable to hypothesise that similar cilia mediated mechanotransduction pathways may be present in tendon. This studentship will test the overall hypothesis that injurious mechanical loading influences primary cilia structure & function, creating a catabolic cell response & leading to the development of tendinopathy. This work is being conducted by Daniel Rowson funded on an EPSRC PhD studentship awarded via the Institute of Bioengineering at QMUL.
Tendon is mechanosensitive, maintaining tissue health in response to applied loads. Overload is a key contributor to the development of tendon pathologies, know as tendinopathies; a range of highly debilitating and increasingly prevalent conditions2-3. However, the mechanisms associated with tendinopathy development remain unclear. Current evidence supports a combined mechanical and cellular pathway, with early structural damage initiating a catabolic cell response. Primary cilia consist of a single microtubule axoneme, 2-4µm in length, which projects into the extracellular environment and regulates fundamental signalling pathways including mechanotransduction, hedgehog (Hh) and Wnt signalling which are important in tissue development, health and disease. Although primary cilia have been identified in tendon, their precise function is unknown. Recent studies of cilia in cartilage indicate that they are essential for tissue development and mechanotransduction mediated matrix synthesis. Furthermore cilia may play a major role in the aetiology of osteoarthritis (OA) through upregulation of Hh signalling8 and alterations in hypoxia inducible factor (HIF) expression (un-published data). Work from Knight’s group also suggests that physicochemical changes in the OA microenvironment, including mechanical loading and inflammatory cytokines, disrupt primary cilia structure and associated signalling pathways. As a tissue damaged through injurious overload, it is reasonable to hypothesise that similar cilia mediated mechanotransduction pathways may be present in tendon. This studentship will test the overall hypothesis that injurious mechanical loading influences primary cilia structure & function, creating a catabolic cell response & leading to the development of tendinopathy. This work is being conducted by Daniel Rowson funded on an EPSRC PhD studentship awarded via the Institute of Bioengineering at QMUL.
