Research
Drug repurposing for treatment of cilia-related pathologies
| Principal investigator: | Martin KNIGHT |
| Co-investigator(s): | Cleo Bishop and Dagan Jenkins |
| Funding source(s): | Queen Mary Innovations |
| Start: 29-09-2020 / End: 01-03-2022 | |
| Amount: £50,000 | |
| Directly incurred staff: | Megan McFie |
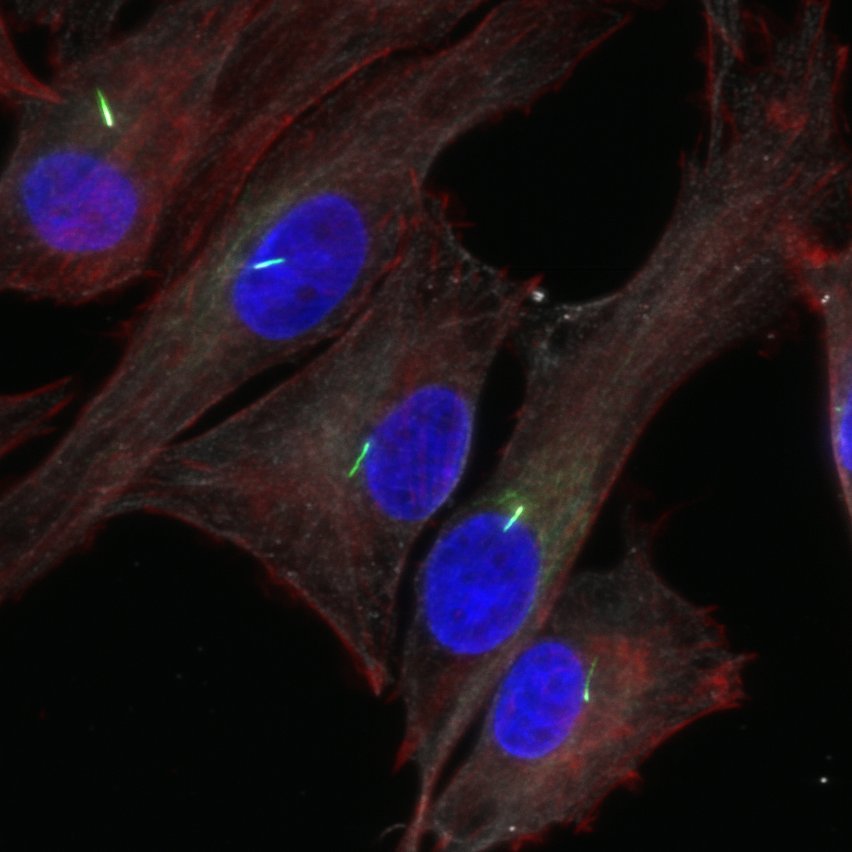
This project funded by Queen Mary Innovations, will identify compounds for the treatment of a rare genetic disease, Jeune Syndrome, that disrupts skeletal formation. The disease is caused by mutation in the genes that regulate primary cilia – the tiny hair-like cellular structures that coordinate a variety of important signalling pathways. The project builds on our recent work conducting a high throughput screen of FDA approved compounds led by PhD student, Megan McFie. This new funding provides £50k to conduct proof of concept studies to test whether hit compounds can correct the genetic defect in primary cilia and prevent life-threatening skeletal malformation.
Prof Knight and collaborators Dr Cleo Bishop and Dr Dagan Jenkins are excited to start this project and hope that it will open the way to new treatment for Jeune Syndrome as well as other conditions associated with disruption of primary cilia. Working with the team at QMI provides valuable expertise on IP and commercialisation pathways to ensure the research has maximum clinical and societal benefit. The team are now looking for industry partners to help develop this potential therapeutic strategy for Jeune syndrome and other related conditions by using cilia modifying compounds identified by this work.

