News
New CRUK/EPSRC research grant: Organ-on-a-chip model to investigate breast cancer bone metastases
12 January 2021

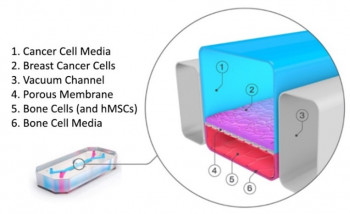
A new research grant using organ-on-a-chip technology to understand breast cancer bone metastasis has started in Prof Martin Knight's bioengineering and mechanobiology research group. The multidisciplinary award is jointly funded by Cancer Research UK (CR UK) and the Engineering and Physical Sciences Research Council (EPSRC).
The project will develop a new organ-chip model of bone metastasis using the Queen Mary+Emulate Organs-on-chips Centre. The researchers will then use this model to examine the role of mechanical loading of bone and bone cell primary cilia in regulating tumour progression.
A new post doc, Dr Joanne Nolan, joins the team led by Prof Martin Knight (SEMS) with Dr Oliver Pearce (Barts Cancer Institute) and Dr Stefaan Verbruggen.
See below for further details on this new research project:
Background
A common site for invasive ductal carcinomas (IDC) metastasis is bone, affecting about 70% of patients. Once metastasis to bone has occurred the five-year survival rate drops from 99% to 29%. How breast cancer metastasises to bone is poorly understood, partly because of the lack of appropriate models. Organ-on-a-chip technology is a new branch of bioengineering which may be used to accurately recapitulate bone metastasis by combining multiple cell types in a microfluidic chip with circulating media and physiological biomechanical forces. However a reliable breast cancer bone metastasis chip is currently not available.
Aims
The aim of this study is to develop an organ-on-a-chip model of bone metastasis in breast cancer and to use this to test the hypothesis that the interaction between cancer cells and osteocytes controls the metastatic niche and tumour development. We hypothesize that this process is regulated by physiological biomechanical loading of bone and osteocyte primary cilia.
Methods
In partnership with the company Emulate Inc., we will develop a specialised organ-on-a-chip model using microfluidic platform technology coupled with optimised biomechanical loading of the different bone cell types in co-culture with breast cancer cells maintained in a 3D matrix. We will characterise this new model and validate it against known chemotherapeutics in terms of phenotypic stability, cancer cell proliferation, migration and invasion and bone formation and remodelling. To test our hypothesis we will examine cancer cell behaviour with and without bone cells and mechanical loading. We will then quantify cancer cell behaviour following treatment with siRNA and pathway antagonists to disrupt osteocyte ciliogenesis and regulation by cancer cells within the chip. Finally we will test the effect of pharmaceutical regulation of osteocyte cilia to identify the potential of novel ciliotherapy for treatment of bone metastasis.
How the results of this research will be used
The project will deliver novel organ-on-a-chip technology for investigating bone metastasis in breast cancer and will be used here to examine the hypothesis that bone cells regulate metastasis in response to mechanical loading and modulated by changes in primary cilia. This is likely to be transferable to other cancers involving bone metastasis and may lead to novel ciliotherapies. The chip may also be modified for use with autologous cells making it suitable for personalised medicine therapies and will provide a scalable platform for testing new cancer therapeutics.
| Contact: | Martin Knight |
| Email: | m.m.knight@qmul.ac.uk |
| Website: | |
| People: | Martin KNIGHT Stefaan VERBRUGGEN |
| Research Centre: | Bioengineering |
Updated by: Martin Knight




