News
Queen Mary partners with the Faraday Institution to power up redox flow batteries
1 June 2022
Queen Mary’s Dr Ana Jorge Sobrido will lead a project to overcome engineering issues that are currently preventing the wide-spread adoption of redox flow batteries (RFBs).
RFBs are emerging as a crucial technology for the transition to renewable energy, since their large capacity can help to stabilise energy grids and store energy from intermittent sources such as wind.
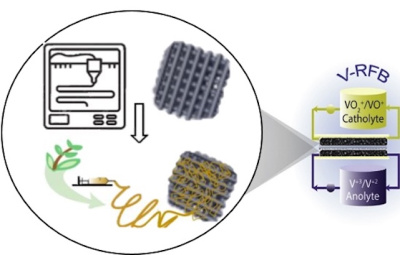
Researchers will combine two flexible, scalable manufacturing methods – 3D printing and electrospinning – to develop an innovative concept of 3D electrodes that will enable optimised mass transport and electrochemical properties. This will be validated by testing a prototype vanadium RFB.
Dr Sobrido, Associate Professor in the School of Engineering and Materials Science, said, "I am absolutely delighted to have been awarded this Seeds Project funded by the Faraday Institution to investigate 3D porous electrodes for redox flow batteries. With collaborators from the UK and from Canada, I will combine the benefits of bespoke 3D-printed electrode engineering with tuneable surface properties of high surface area electrospun fibres. This will produce controlled microstructural properties that can optimise mass transport through the electrode, and enable increased surface area over commercial carbon felts. This project represents a fantastic opportunity to advance the field of redox flow batteries and I am grateful to the Faraday Institution for their support."
Dr Sobrido expects that the materials developed could also find be useful in other battery technologies, fuel cells and electrolysers where engineered electrode structures would have mass transport performance benefits.
Professor Pam Thomas, CEO of the Faraday Institution, said: “We’re absolutely delighted that with the launch of this project that QMUL has joined the Faraday Institution research community. The Faraday Institution is the UK’s independent institute for electrochemical energy storage research, skills development, market analysis, and early-stage commercialisation. It brings together research scientists and industry partners on projects with commercial potential that will reduce battery cost, weight, and volume; improve performance and reliability, and develop whole-life strategies including recycling and reuse. The Faraday Institution now has 27 university partners and 500+ researchers looking to address these challenges – very much a national programme."
The award is one of 16 small, fast-paced, focused projects in areas not covered within the Faraday Institution’s existing battery research portfolio. In total, 14 universities are involved with the seed projects, which will run for 12 months and represent a £2 million investment in research by the Faraday Institution. The funding round was highly competitive; it was oversubscribed by four times.
| People: | Ana JORGE SOBRIDO |
| Research Centre: | Sustainable Engineering |




