News
Why does graphite need water to be a lubricant? The answer isn't what we thought
18 March 2026
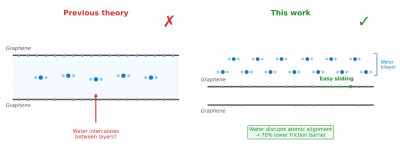
New research overturns the long-held theory that water slips between graphene layers to lubricate graphite — instead, it sits on the surface and disrupts the perfect atomic alignment that causes friction.
In the 1940s, the US Air Force had a problem. The graphite brushes in electrical motors on high-altitude aircraft were wearing out far too quickly. By the 1970s, NASA confirmed the issue: graphite simply stops working as a lubricant in the dry vacuum of space. Clearly, atmospheric water was essential — but why?
For decades, the leading explanation was intuitive: water molecules must slip between the graphene layers that make up graphite, enabling them to slide past one another. It was a neat theory, but the experimental evidence has been contradictory. X-ray diffraction measurements show no change in the spacing between graphene layers at different humidity levels — suggesting water stays out. Yet recent Raman spectroscopy experiments on bilayer graphene show clear signatures of water getting in.
A new study published in Physical Review Materials by Dr Yiwei Sun from the School of Engineering and Materials Science (SEMS), together with Professor Sir Colin Humphreys (SEMS), Professor David Dunstan (School of Physical and Chemical Sciences), and collaborators at Montanuniversität Leoben in Austria, now resolves this decades-old puzzle — and the answer challenges conventional thinking.
The 80% threshold
Using density functional theory (DFT) calculations, the team first showed that the interlayer bonding near graphite's surface is indeed about 6% softer than in the bulk — supporting the idea that water might find it easier to enter at the surface. But when they ran molecular dynamics simulations to test whether this softening actually allows water in, the answer was firmly no.
"We found that water can only intercalate between graphene layers when the van der Waals interactions are reduced by over 80%," said Dr Sun. "The 6% surface softening doesn't even come close. It's like saying a door is slightly ajar — but you'd actually need to remove most of the wall."
It's not between the layers — it's on the surface
Instead of slipping between layers, the simulations reveal that water forms a remarkably stable two-atomic-layer structure on the graphene surface. This bilayer persists from room temperature up to nearly 380 K, and remains intact even on realistically curved graphene surfaces — the kind found at asperity contacts in real machinery.
The key insight is what this surface water does to friction. Without water, graphene layers stack in perfect atomic registry: as one layer slides, every atom hits an energy barrier at the same time, like trying to push an entire crowd through a turnstile simultaneously. The surface water layer disrupts this perfect alignment — a phenomenon called incommensurability — so that while some atoms are going uphill, others are going downhill, and the energy barriers largely cancel out.
The numbers are striking: the sliding energy barrier drops from approximately 72 mJ/m² for dry graphene-on-graphene to just 22 mJ/m² when water is present — a 70% reduction.
Why MoS₂ doesn't need water
This mechanism also explains a textbook puzzle: why does MoS₂ lubricate perfectly well in dry conditions while graphite fails? The answer lies in the strength of the interlayer bonds. MoS₂ has fundamentally weaker van der Waals interactions and wider layer spacing (6.2 Å versus 3.35 Å for graphite). Its layers achieve incommensurability easily through minor thermal fluctuations alone — no water needed.
"It's a design principle for layered lubricants," said Dr Sun. "You either need inherently weak interlayer forces, as in MoS₂, or you need something — like water — to break the atomic-scale alignment. Graphite happens to need the latter."
Relevance to materials science and beyond
The findings have implications beyond graphite lubrication. Understanding how molecular adsorbates modify friction at the nanoscale is relevant to the design of coatings, nanoelectromechanical systems, and any application where layered materials slide under ambient conditions. The work also provides a quantitative framework — the 80% vdW reduction threshold — that can be applied to assess intercalation feasibility in other 2D material systems.
The study combined DFT calculations for interlayer stiffness, reactive molecular dynamics simulations using ReaxFF potentials, and a reinterpretation of published experimental data on graphene friction — demonstrating the value of integrating computational and experimental approaches to resolve longstanding materials puzzles.
| Contact: | Yiwei Sun |
| Email: | yiwei.sun@qmul.ac.uk |
| Website: | |
| People: | Yiwei SUN Colin HUMPHREYS |




